 
All the elements are arranged in horizontal rows and columns (called periods). A periodic table is called so because of how the elements are arranged. 
It used to correctly predict elements that hadn't been discovered.Īns. The periodic table reflects its creator love for card games. Scientists before used battery polarity to weigh the elements. Facts About the Periodic Table of Elements are mentioned below:ĭmitri Mendeleyev is the founder of the modern periodic table. What are some fun facts about the periodic table?Īns. 58.9) is placed before Ni (58.7) and Ar (39.9) is placed before K (39). The order of increasing atomic weights is not strictly followed in the arrangement of elements in the periodic table. It is not clear to which group lanthanides and actinides belong to.Īlthough there is no resemblance except valency of subgroups A and B, they have been put in the same group. No separate positions are given to isotopes. It has been placed in IA and VIIA groups because of its resemblance with both the groups. 
Where this gap was more, the gaps were left in the periodic table. Mendeleev calculated it 2 × 4.5 = 9.ĭiscovery of new elements – In Mendeleev's periodic table two consecutive members differs by two or three units in the atomic weight. Atomic weight of Be was calculated to be 3 × 4.5 = 13.5 by considering its valency 3. Atomic weight = Valency × Equivalent weight = Group number × Equivalent weight.Ītomic weights of elements were corrected. Eka boron (Silicon) Eka silicon (Germanium), Eka aluminum (Gallium) Eka Maganese (Technitium)Ītomic weights of elements were determined with the help of periodic table. When the properties of elements did not correspond to what is expected of the group they were named by prefixing Eka to the preceding element e.g. Leaving gaps for the then undiscovered elements He excluded certain elements and assigned them a separate independent position. Mendeleev's periodic table was published in the German Journal of chemistry in 1869. Only about half of the elements we know now were known during his time, and most of the information about all elements was inaccurate. Mendeleev modeled his periodic table based on increasing atomic mass, while the modern periodic law is based on increasing the order of atomic numbers.Īlthough Mendeleev's periodic table was based on atomic weight, he could predict the properties and discovery of specific elements. Mendeleev's periodic law differs from the modern periodic law in one significant aspect. Thus, Mendeleev is given sole credit for his development of the periodic table.ĭimitri Mendeleev, who is widely regarded as the father of the periodic table, came up with the first iteration of the periodic table that is similar to the one we use now. Dimitri Mendeleev presented his periodic table based on the findings of some scientists before him, such as Antoine-Laurent de Lavoisier and John Newlands. The modern periodic table we use now is a new and improved version of certain models put forward by scientists in the 19th and 20th centuries. The first 94 periodic table elements occur naturally, while the remaining 95 to 118 have only been synthesized in laboratories or nuclear reactors. Therefore, as the energy level of an atom increases, the number of energy sublevels per energy level increases. While elements in the same period will have increasing order of valence electrons. Therefore,Įlements in the same group that have the same configuration of valence electrons and, therefore, similar chemical properties. The elements are arranged from left to right and top to bottom in increasing order of atomic numbers. They are arranged in a tabular arrangement where a row is a period, and a column is a group. 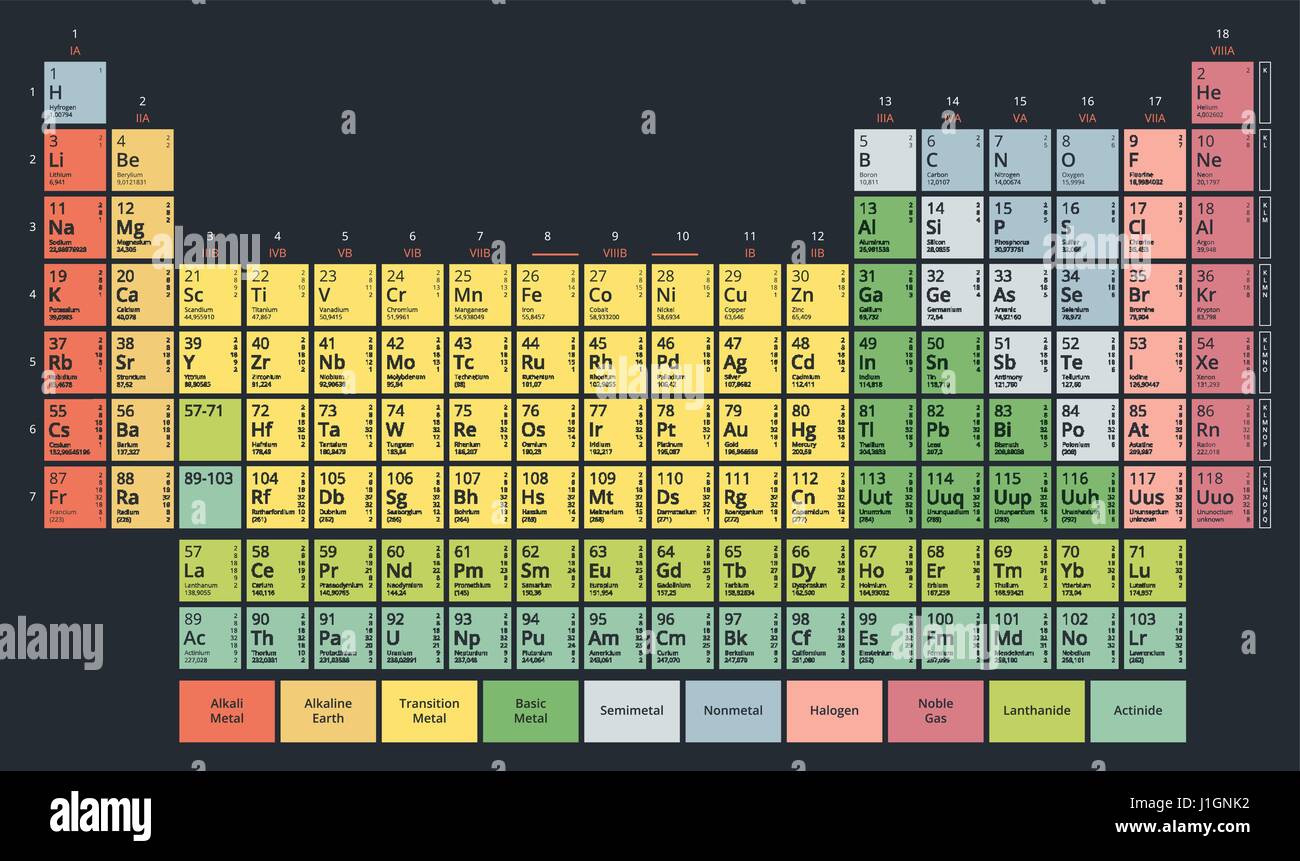
A periodic table is an arrangement of all elements known to man according to their increasing atomic number and recurring chemical properties. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
